Galvanic Corrosion
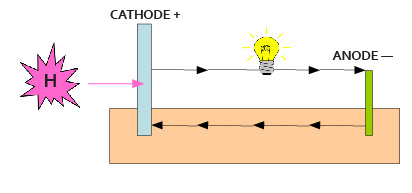
When combining two different metals in the presence of an electrolyte (a fluid that conducts electricity) hydrogen is absorbed at the cathode and a current flows; one of the metals, the ‘anode’, is attacked.
This is the basic mechanism for simple batteries.
The size of the cathode relative to the anode controls the power generated and the corrosion level at the anode.

GALVANIC ATTACK ON ROOFING & CLADDING MATERIALS
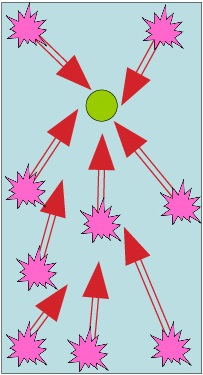
BAD design for fastening
Large cathode absorbs lots of hydrogen and drives corrosion against small anode; cathode is ‘protected’.
‘Sacrifical anode’ effect = fastener at risk.
Stainless fastner [cathode] Aluminium sheet [anode]
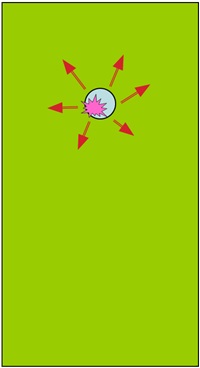
Good fastner protection
Area Rule Still Applies*
GOOD design for fastening
Small cathode attracts small level of hydogen and so has weak effect against large anode.
Cathode (fastner) is ‘protected’ and little corrosion effect on large anode
Stainless fastner [cathode] Aluminium sheet [anode]
Good fastner protection
Area Rule Still Applies*
You can keep a copy of this information by downloading the PDF here.





Leave A Comment
You must be logged in to post a comment.